Modern hospitals do not just maintain water systems. They manage risk.
A Water Management Program is often treated as a requirement or a policy, something that needs to exist for compliance purposes. In reality it is much more than that. It is an operational system that must function daily, adapt over time, and integrate into the way a hospital actually runs.
This is where many programs fall short. The issue is not the design of the program, but how it is executed. A Water Management Program should not sit on paper. It should guide real actions, real decisions, and real outcomes.
To understand how to make that happen, it helps to look at the program as a system you can actively run.
When executed properly, a Water Management Program follows a defined sequence. Here's what that looks like:
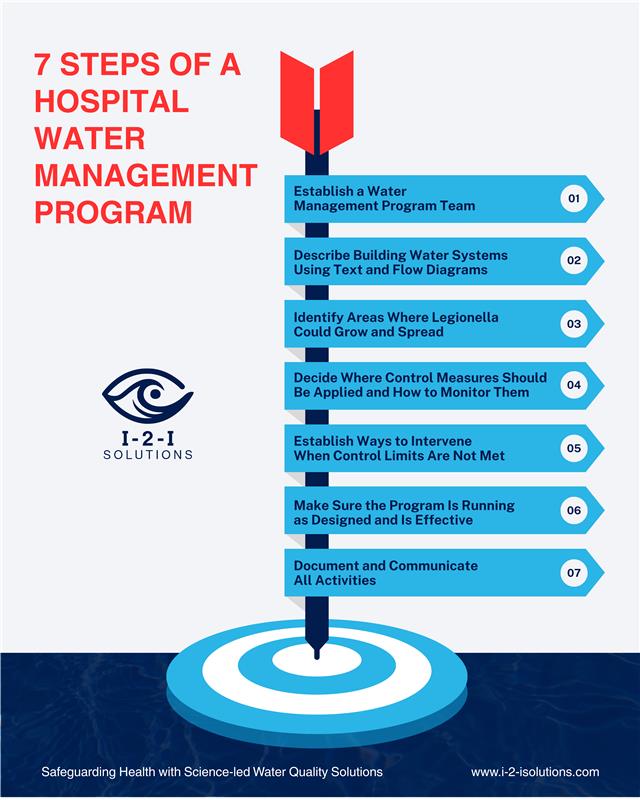
Step 1: Assemble The Right Team
A Water Management Program does not belong to a single department. It requires coordination between facilities, infection prevention, environmental health and safety, and clinical leadership. What matters here is not just representation, but accountability. Each role should be tied to clear responsibilities, from reviewing results to initiating corrective actions and documenting decisions. This is what defines how decisions are made in practice.
Step 2: Map Your Water System
Hospitals have complex water systems that include potable water, cooling towers, storage tanks, and various outlets throughout the facility. Mapping the system means understanding how water flows, where it may stagnate, and which areas present higher risk. This map becomes a working tool that supports sampling, monitoring, and intervention rather than just a static diagram.
Step 3: Identify Risk Points
Not every part of the system carries the same level of risk. Certain conditions such as warm temperatures, low disinfectant levels, stagnant water, and the presence of biofilm create environments where bacteria like Legionella can grow. This step focuses attention on the areas that matter most, allowing teams to prioritize resources effectively.
To better understand how Legionella spreads through building water systems and which environments are most vulnerable, see our detailed breakdown of where people are most likely to be exposed to Legionella.
Step 4: Define Control Measures
Once risks are identified, there must be clear ways to manage them. This can include temperature control, flushing protocols, disinfection strategies, and equipment maintenance. For these measures to be effective, they need to be measurable, assigned to specific individuals, and built into regular schedules. If a control measure is not part of a routine, it is unlikely to be carried out consistently.
Step 5: Establish Monitoring Procedures
Control measures only work if they are consistently tracked. Monitoring may include checking temperatures, measuring disinfectant levels, and verifying system performance. The purpose is to confirm that the system is operating as intended. To be effective, monitoring must align with existing workflows rather than exist as a separate or occasional task.
Step 6: Validate Through Testing
Environmental testing plays a critical role in confirming whether control measures are working. Testing provides early detection of potential issues and offers data that can guide decision making. For testing to be meaningful, it must be conducted regularly, strategically planned, and properly interpreted. This is what turns assumptions into evidence.
I-2-I Solutions supports all kinds of healthcare facilities with science-backed Legionella and Dental Water testing with clear results, expert guidance, and the documentation your facility needs to move forward confidently.
Our laboratory practices align with standardized methods such as ISO 11731:2017, supporting accurate and reproducible detection of Legionella in water systems. We perform culture testing in-house, allowing for greater control, consistency, and confidence in identifying viable organisms that pose real risk.
Step 7: Act and Continuously Improve the Program
The Water Management Program is not static. When monitoring or testing reveals a problem, the response must be immediate, documented, and effective. Actions can include flushing systems, applying disinfection, or upgrading equipment. Just as important is reviewing outcomes and refining the program over time. Strong programs evolve as conditions change.
Many Water Management Programs do not fail because they are missing components. They fail because responsibilities are unclear, tasks are not integrated into daily operations, and data is not used to guide decisions. When that happens, the program exists in theory but not in practice.
A well-functioning program goes beyond compliance. It creates visibility into system performance, builds confidence in decision making, and protects both patients and staff. Water safety in hospitals is not theoretical. It is operational and continuous, and it must work every day.
If you are evaluating your current program, the most important question is not whether you have one. It is whether it is something your team can run. Just as important, though, is whether you have chosen the right laboratory to support it.
The accuracy, consistency, and credibility of your results depend heavily on the lab behind them. A qualified laboratory doesn't just process samples, it ensures that your decisions are based on reliable, defensible data. That's why working with an ISO/IEC 17025 accredited lab that follows recognized standards and participates in independent proficiency testing such as the CDC ELITE Program is critical to maintaining a program that is both effective and compliant.
To learn how to choose the right laboratory for your water management program, read our guide: 5 Steps to Evaluate a Legionella Testing Laboratory.
Why I-2-I Solutions Supports High-Performing Water Management Programs
At I-2-I Solutions, our approach is built around the same principles used to evaluate high-performing Legionella testing laboratories. We are ISO/IEC 17025 accredited and emphasize independent performance verification through participation in recognized proficiency testing programs, including the CDC ELITE Program and ISO 17043-accredited programs such as those provided by NSI Lab Solutions, which are required to maintain ISO 17025 accreditation. This demonstrates our commitment to nationally recognized standards for laboratory competence and quality management.
